TiGER
Our CRISPR workflow increases the speed and efficiency of your plant breeding
HRB employs CRISPR-based gene editing for specific trait development. CRISPR can be used to target highly specific pieces of DNA while leaving the rest of the genome intact. This allows for very fast improvement of already carefully bred organisms. Our end to end workflow allows you to target different traits that could bring value for your business. We target both agronomic and consumer traits, examples include:






Plant breeding with CRISPR
The key strength of CRISPR-based breeding is that it allows for faster and more targeted development of crop varieties. After several years of development, CRISPR has become a mature enough technology to deliver the plants that we need for the future, in a safe and controlled manner. Where similar type of crop adjustments would traditionally take between seven and ten years (if possible at all), they can now be done within two to four years.
To change a specific DNA sequence, the CRISPR system relies on a DNA cutting enzyme (endonuclease) that is guided to a target sequence using a guide RNA (gRNA). By cutting and later repairing the DNA, the sequence can be changed in a desired way. 3 main steps are necessary:
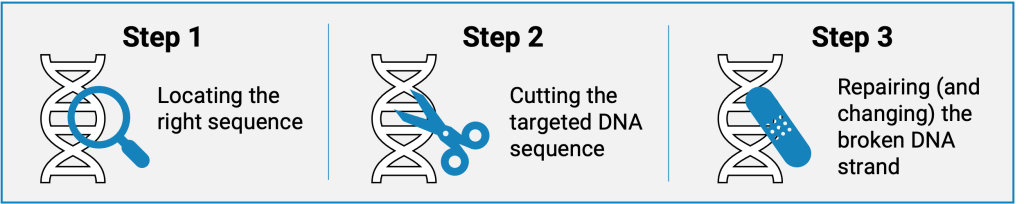
The gRNA drives the first step of CRISPR: finding the right DNA sequence on the genome of a cell. One part of the gRNA’s sequence can be designed to fit a complementary sequence in the DNA of the genome. This then guides the cutting enzyme to any location in the genome where this recognizable motif is present. If the chosen sequence is unique enough in the genome, this location will be the only site targeted.
Upon binding of the gRNA to the DNA of the genome, the associated endonuclease initiates the second step of the CRISPR system: cutting the targeted DNA motif. It cleaves both DNA strands so that open ends are created in the genetic material.
In the final CRISPR step, the cell itself performs the actual genome editing. DNA breaks occur naturally all the time. As these are potentially damaging, the cell has its own repair systems in place. We make use of these natural repair systems to fix and edit the DNA we intentionally cut.
Common challenges of CRISPR plant breeding
Even though the benefits of applying CRISPR in molecular breeding are clear, the technology is often held back by various challenges. The cost and complexity of projects can rise quickly, while many CRISPR workflows face regulatory issues. The main challenges associated with CRISPR editing in agriculture include:
High licensing costs
Regulatory issues
Long project lead times
High complexity
How does the TiGER workflow address these issues?
For a wide range of species, our TiGER workflow overcomes CRISPR bottlenecks to rapidly deliver top-quality products that can be directly introduced into the market development pipeline. In this way, we tackle the most common challenges associated with CRISPR gene editing.
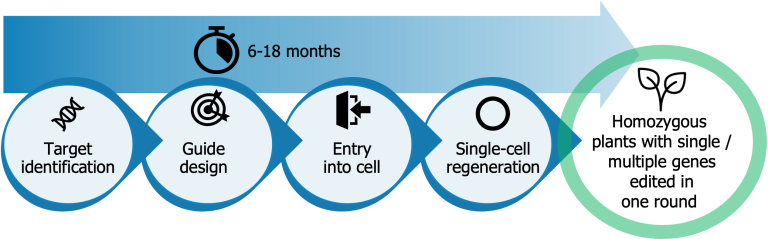
Our proprietary TiGER workflow yields a genetically uniform edited organism in 6-18 months, offering benefits compared to traditional approaches:
- Transgene-free delivery protocols without the use of foreign DNA, which limits regulatory issues;
- MAD7 nuclease, a commercially more attractive option than Cas9, with high editing efficiencies to minimize costs for edit screening;
- Proprietary guide design software to target single or multiple genes in one editing round while minimizing chances of off-target edits;
- Single-cell protoplast regeneration to avoid edited chimeras. Single-cell protoplast regeneration to avoid edited chimeras. Even for recalcitrant species difficult to handle in vitro, we have achieved methods to regenerate these. In addition, we are developing a nanoparticle-based transfection-delivery method.
By applying our workflow, you can speed up the development of plant breeding, even in comparison to other CRISPR approaches.
What our clients say

Read about our proprietary CRISPR workflow in the whitepaper
Our TiGER workflow enables molecular breeding in an end-to-end fashion, providing innovative solutions to the typical barriers encountered in CRISPR breeding projects. Read about our step by step approach by downloading our whitepaper.